These are salt fingers. The eerie fingerlike structures are caused by sinking blobs of warm, salty water interleaving with rising blobs of cold, fresh water.
This is a thermohaline staircase. Thin sheets of well-mixed water stack on top of each other to create a stepped vertical profile.
But these two seemingly different oceanic phenomena have something in common. They are both caused by the same process, double diffusion.
Diffusion means the spreading out of a certain property or particles. When you fart on a plane and then you sit really still hoping no one notices but the stank still manages to escape and terrorize everyone in a 3-row radius, that is diffusion. Similarly, diffusion occurs in the ocean. But fart particles in the ocean don’t spread out as fast as when you are on a plane because diffusion in water is much slower than diffusion in air (technically farts don’t actually diffuse in water because they are mostly air and just rise to the surface. You can test this by farting in your bathtub, for science of course). In fact, diffusion in water is so slow that other transport mechanisms like mixing and currents would spread your aquatic fart particles throughout the seven seas. But when these other oceanic processes are minimized, diffusion is the boss!
Diffusion in sea water also doubles your pleasure and your fun. Double because sea water has two characteristics that diffuse, salinity and temperature. What makes double diffusion so special is that heat and salt don’t spread out at the same rate. A warm, salty blob of seawater will lose heat nearly 100 times faster than it will lose salt, so it will become colder long before it becomes fresher. And this is what leads to an ocean full of staircases and fingers.
Salt fingers form when warm, salty water overlies cold, fresh water. Initially, the warm, salty water floats on top because it is slightly less dense than the cold, fresh water*. As the warm, salty water loses heat faster than it loses salt, it becomes denser. Similarly, the cold, fresh water is becomes less dense as it warms but doesn’t become much saltier. This leads to the water being unstably stacked. Eventually, the warm, salty water becomes so dense it sinks while the cold, fresh water floats. This happens rather quickly, and in a spectacular fashion, creates the vertical plumes of water known as salt fingers.
To really appreciate the beauty of salt fingers, you need to watch them form. You could do this with a little mini-experiment at home (which I tried and failed at today, but will try again tomorrow) or just check out the simulation I pasted below.
Thermohaline staircases form in the opposite scenario: cold, fresh water overlies warm, salty water. Heat and salt diffuse upwards. The lower layer gets a little colder but stays denser, while the upper layer gets a little warmer and lighter. Unlike salt fingering this creates a stable scenario, where the densest water is at the bottom, so no crazy plumes are formed. But the upward diffusion of heat still causes mixing over small vertical scales, which over time creates the layers that make up the thermohaline staircase.
The conditions to create thermohaline staircases can be found all over the globe. But they can easily be destroyed due to turbulence which muddles the fine layers together. But there is one ocean where thermohaline staircases reigns supreme, the Arctic. Here sea ice and stable stratification quells the processes that typically mix oceans at lower latitudes, allowing the staircases to persist. At the surface, there is a cold, fresh layer of water due to ice melt and terrestrial river runoff. Just beneath is a layer of warm and salty water which originates in the North Atlantic and circulates around the Arctic. Staircases are formed between these layers. But staircases can also be found in the Caribbean Sea driven by warm evaporation at the surface and beneath the salty tongue of the Mediterranean outflow in the Atlantic.
Thermohaline staircases can even be found in bodies of freshwater such as Lake Kivu on the border of Rwanda and the Congo, where seeps due to volcanic activity create a layer of warm, salty water under the cool, fresh lake water. But these staircases also have a sinister side. The stability that allows the formation of staircases also traps dangerous gases such as carbon dioxide and methane at the bottom of the lake, which could potentially explode. Similar “lymnic explosions” have been documented at two other lakes, with deadly consequences.
In the ocean, double diffusion is not a hazard nor is it just a curious phenomena that make cool images. When scientists first observed the extent over which double diffusion occurred, it was thought double diffusion could be a major source of mixing in the ocean, both laterally and vertically. Since the strength of global circulation is sensitive to how much vertical mixing occurs, double diffusion could control some aspects of it. As with all things new and novel, many expeditions were conducted and many models were run, but the importance of double diffusion on global circulation is still unknown.
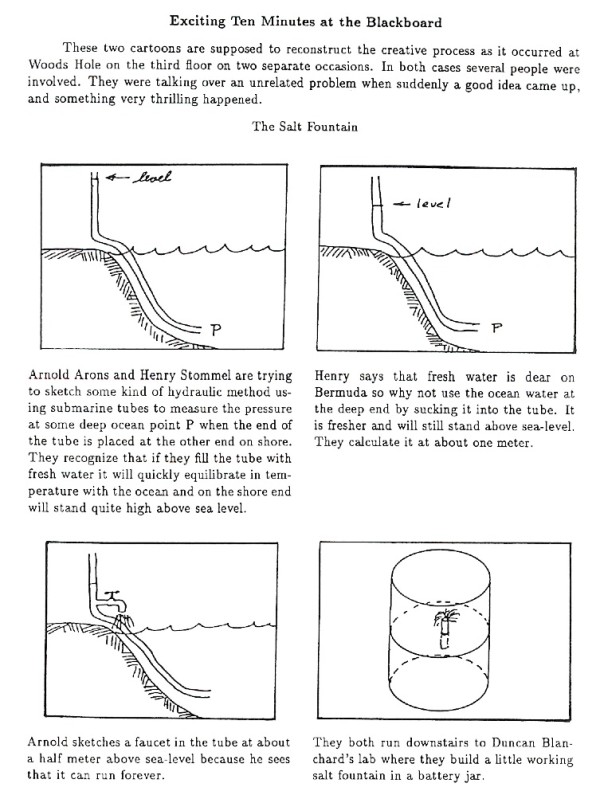
Finally, I leave you with this little nugget of joy. This is the illustrated description of the discovery of double diffusion from the Collected Works of Henry Stommel. Because no one can resist impromptu science.

*Sea water density is determined by its temperature and salinity. Cold water is usually denser than warm water and salty water is usually denser than fresh water. In a typical ocean, this holds true as light warm and fresh water is found at the surface while heavy cold and salty water is found deeper. In the case of salt fingers, temperature has a greater effect on density than salinity. Therefore, the cold, fresh water is denser than warm, fresh water.
REFERENCES:
Carpenter, J. and M.-L. Timmermans, 2012. Temperature steps in salty seas. Physics Today, 65(3), 66 (2012); doi: 10.1063/PT.3.1485.
Hogg, N. G., and R. X. Huang. “Collected Works of Henry Stommel.” American Meteorological Society, Boston (1996).
Ruddick, Barry, and Ann E. Gargett. “Oceanic double-infusion: introduction.“Progress in Oceanography 56.3 (2003): 381-393.
Timmermans, M.-L., Toole, J., Krishfield, R. and Winsor, P., 2008. Ice-Tethered Profiler observations of the double-diffusive staircase in the Canada Basin thermocline. Journal of Geophysical Research, 113, C00A02, doi:10.1029/2008JC004829.
Share the post "Ocean staircases and salt fingers, the curious case of double diffusion"






but, Dr. Martini, what about all the air vents on the plane…don’t they help spread out the fart smell…aka advection? This analogy is awesome, but I think it begs for a follow-up clarification on differences between advection and diffusion.
Good luck getting your demo to work…that one can be tricky.
This is all true. Advection is the transport of matter or heat by fluid flow. So the air vents will advect the fart around. Just like currents advect around sediment, sea beasties and heat.
But in all this talk of air vent advection, let us not forget the primary source of fart advection. The force needed to expel such a fart from your butt.