
The deep-sea sponge Monorhaphis chuni (Hexactinellida) has the world’s largest known biosilica structure! A silicate spicule that can grow up to 3 meters long. That’s at least a meter longer than you! I don’t really know how to construe to enormity of such a structure. I could make it so you have to scroll down this post 3 meters, but that would just be annoying.
Müller and colleagues carefully studied the formation of these giganto-spicules and found formation was helped along the way by silicatein-related proteins. Silica is not a common element in the ocean. Though rarer at the surface it increases in concentration 10-fold as you go deeper. The ability to scavenge this rare element and incorporate into a biostructure is in itself a fantastic feat – an energetically expensive one at that! Now extend that from the micrometer to millimeter scale over time to 3 meters.
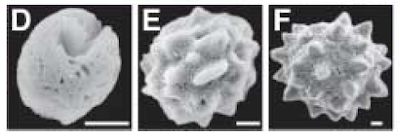
This study is an excellent exercise in integrative biology merging biochemistry, histology, genetics, morphology and systematics. They determined there were different chemical layers to the spicules, including collagen and the silicateins (potentially a first for the Hexactinellida). They concluded:
Based on the data gathered here, we suggest that, in the Hexactinellida, the growth of the spicules is mediated by silicatein or by a silicatein-related protein, with the orientation of biosilica deposition being controlled by lectin and collagen.
Keeping with the theme of sponge skeletons, but moving away from silica-based to carbonate-based, another study by Jackson and colleagues in Science used an approach called Palaeogenomics to determine the role of precursor α-Carbonic Anhydrases (α-CA’s) in calcareous skeleton formation. Palaeogenomics uses modern techniques, such as gene and protein expression and phylogenetics, on extant organisms in combination with knowledge of their evolutionary history. α-CA’s have evolved through several gene duplication events in the Metazoa for a variety of physiological purposes:
The chemical reaction [CO2 + H2O ⇆ HCO3− + H+] functions in processing metabolic wastes, regulating pH, fixing carbon, and transporting ions across organic membranes. The metalloenzyme carbonic anhydrase is pivotal to these processes by catalyzing this reaction approximately 1 million fold.
This is a very important enzyme with a diverse set of functions cascading down throughout the Metazoa. The aim of this paper was to see what α-CA’s may have looked like in the last common ancestor to the Metazoa (LCAM). Sponge genomes are great to look at for these types of questions because of their basal position on the animal tree of life.
The authors determined the α-CA enzyme is used in biocalcification of the Demosponges. The sponges (and presumably biocalcification) radiated in the Cambrian Explosion, 520-540- million years ago. The LCAM most likely used the a-CA enzyme for a similar purpose. And in fact we see this feature, biocalcification, presevered in several protostome and deuterostome taxa. As with any well written Science paper, there is a succinct final paragraph concluding their results:
From our data we infer that a core molecular toolkit capable of catalyzing the production of HCO3− (and ultimately CaCO3) was present in the first metazoans and included an α-CA. Subsequently, various metazoan lineages inherited this toolkit and have added to and elaborated upon its key elements to guide, enhance, and inhibit the deposition of CaCO3 in the spectacular variety of ways we see today.
Two well-written sponge papers with important evolutionary conclusions. What more could you ask for?
Jackson, D., Macis, L., Reitner, J., Degnan, B., & Worheide, G. (2007). Sponge Paleogenomics Reveals an Ancient Role for Carbonic Anhydrase in Skeletogenesis Science, 316 (5833), 1893-1895 DOI: 10.1126/science.1141560
Müller, W., Eckert, C., Kropf, K., Wang, X., Schloßmacher, U., Seckert, C., Wolf, S., Tremel, W., & Schröder, H. (2007). Formation of giant spicules in the deep-sea hexactinellid Monorhaphis chuni (Schulze 1904): electron-microscopic and biochemical studies Cell and Tissue Research, 329 (2), 363-378 DOI: 10.1007/s00441-007-0402-x
This was originally posted by me on July 21, 2007 at The Other 95%.
Share the post "Huge Silicate Sponge Spicules and the Evolution of Calcification"





4 Replies to “Huge Silicate Sponge Spicules and the Evolution of Calcification”
Comments are closed.